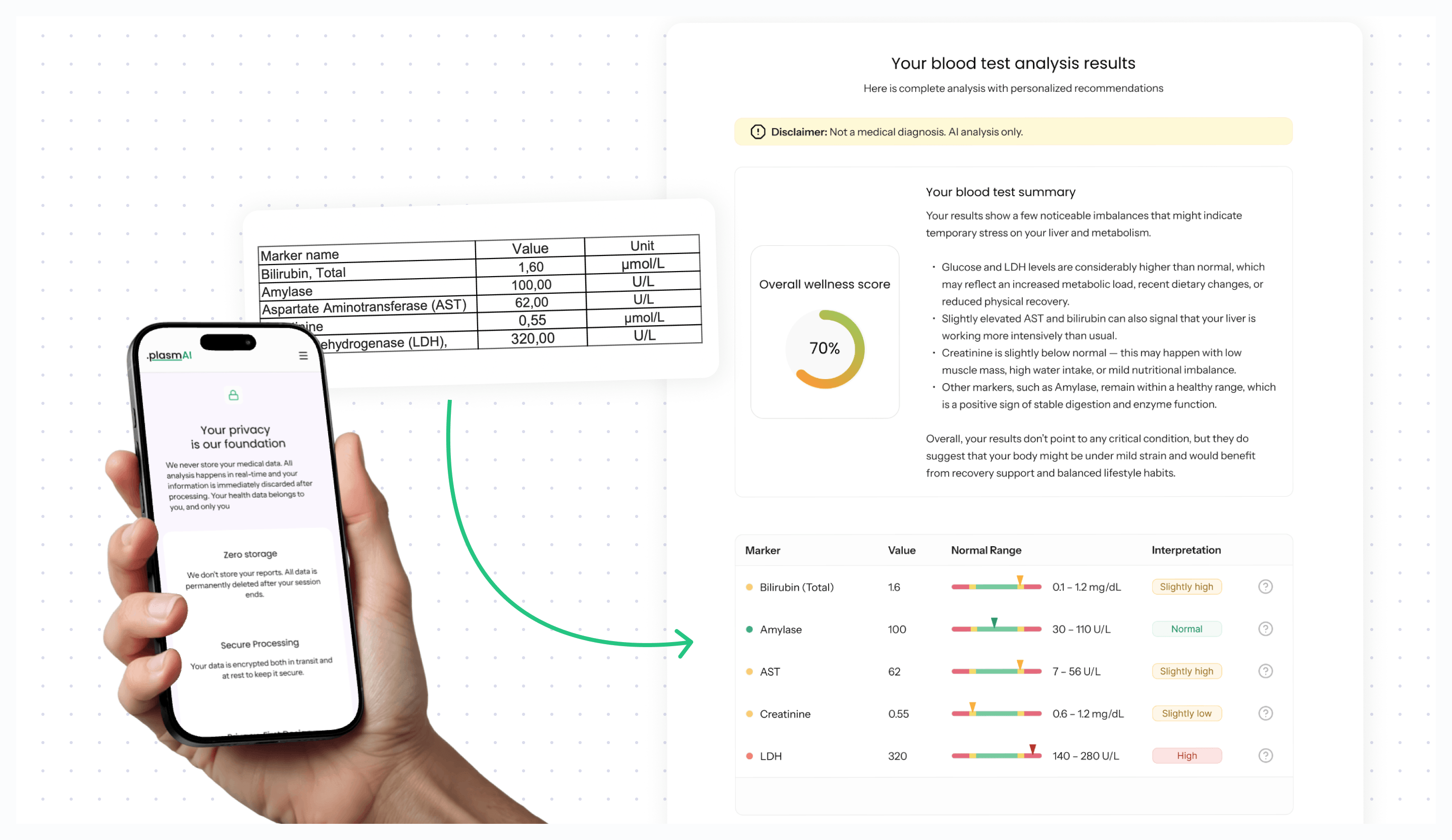
Software development with compliance-ready design
ZenBit Tech had an idea to help patients understand their lab results through AI analysis. The technology extracs biomarkers, AI interprets results and a user-friendly interface delivers clarity.
But medical data is in the most regulated categories of information and any product using it must be compliant before it ever reaches users.
Before go to market, ZenBit Tech needed to prove compliance with:
🇪🇺 GDPR (European Union) for patient consent, lawful basis for processing, data minimization, retention policies
🇬🇧 UK GDPR + Data Protection Act 2018 (United Kingdom) for UK-specific data protection rules, DPIAs, ICO oversight, cross-border transfer safeguards
🇨🇭 FADP / nFADP (Switzerland) for enhanced transparency, stricter consent requirements, data subject rights, cross-border data transfer controls
🇺🇸 HIPAA (United States) for protected health information handling, audit trails, breach notification, Business Agreements
It means you hire experts in every market, wait for slow approvals and build compliance by hand. Yet you still worry you missed something.
So the biggest risk wasn’t AI accuracy, it was data responsibility. Storing patient lab results would immediately make ZenBit Tech a medical data controller. It’d increase legal exposure, audit scope and operational complexity. So, without compliance ready solution, even the best technology product can’t enter the market
And we created architectural background with compliance ready decision in mind — the white label platform for patients without storing the data and API for existing solution with storing through third party partners

Dmitry Broshkov
Software developer
Founder ZenBit Tech